Two important topics in APES are combined for this 5E–Serial dilution Lab and LC50 Lab. The lab papers needed for this lab can be found on this google drive folder.
Engage: Flint, Michigan
This Engage is a short case study about lead exposure for students. Most students have heard of Flint, Michigan and its water quality problems so this adds to previous knowledge (a key component of Engage). The video piques their interest and then we discuss our own drinking water and how this problem would not occur here, because we are a newer community that does not have lead pipes. We also discuss how older cities with lead pipes can prevent lead leaching by using an additive in the water. Click to read this article explaining more about how lead gets into drinking water.
Explore: Serial Dilution
Students in AP® Environmental Science need to understand how toxins can still be prevalent in very small amounts. The best way to do this is for students to do a serial dilution. (Note: This activity is courtesy of Dan Hyke from the APSI, I attended in 2006. I have altered it and combined with LD-50 for this 5E)

As students walk through the procedure, fill in their data charts and answer the guiding questions, they hopefully will come up with the concept on their own–toxins can still be present even in very small amounts such as parts per million (ppm), parts per billion (ppb) and parts per trillion (ppt). Materials needed are: (Click on an underlined item for a link to the product)
- Well plate
- Plastic Pipet
- Beaker for tap water
- Toothpicks
- Dropper bottle of a dye/stain/coloring such as a Food Grade Dye like FD&C Red Dye #40 Do not use regular food coloring as it dilutes much too quickly
Students use 3-4 drops of the red dye solution in the first well. Then they fill the other wells with 9 drops of tap water. After that, they drop one drop from the previous well and stir.
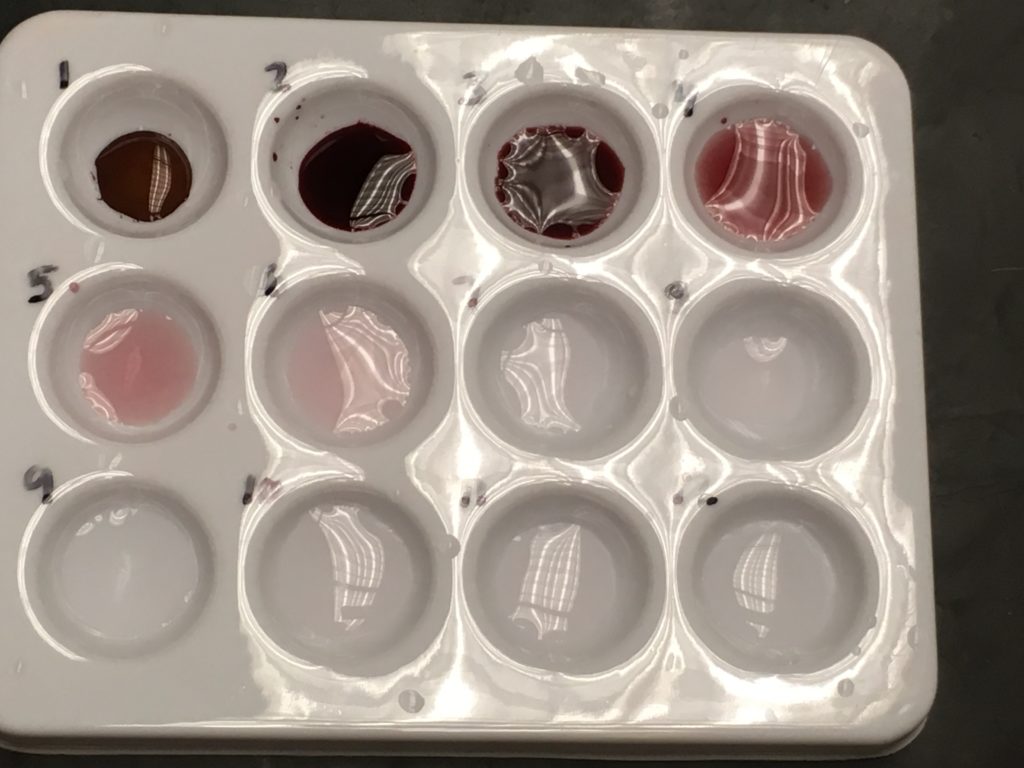
Students are often concerned when their solution is clear by well 7. I tell them that’s normal and they are to still make the transfers. They need to move the molecules in order to understand the point of the lab.
Students creatively identify shades of red and pink and learn about ppm, ppb, and ppt on their data sheet. Some students will need help with these circles. I tell them that “their brains will hurt” today.
Explain: Student Sense-Making
In a good 5E, students should be able to develop the concept you want them to on their own. Developing good guiding questions is your job as the teacher to lead them to it. For this lab, students should make a CLAIM or a STATEMENT that “Substances can be in water even if you cannot see it, smell it or taste it.
Elaborate: LC50 Lab
We use the results from the salinization Lab to do LC-50. Students bring their salinization labs back to class and we collect class data. (Students save all work in their APES binder) This time, however, we want “opposite” data–the number of seeds that DIED, instead of the number that germinated.
Students fill in the chart for their group’s data and then all students copy class data. I often make a chart on the board for groups to fill in.

From there, using a document camera or on the board, teach students how to find the LC-50 by drawing a line from 50% on the y axis to where it hits the dose-response curve and then down to the x axis. Read the concentration that kills 50% of seeds. If you need help with this, I made this video for absent kids that may help you understand how to do this lab:
Evaluate
This 5E does not have its own Evaluate–rather students are assessed for these skills and knowledge on their next exam.
* AP® is a trademark registered and/or owned by the College Board which was not involved in the production of, and does not endorse this site.


5 thoughts on “Toxins 5E Lab”